ENDOFIS - Banco de Patentes
Banco de patentes
Comité Tech Transfer. Webinar: TESTIMONIALS “De la investigación al emprendimiento”
Destinado: Comunidad investigadora. El contexto actual derivado de la situación pandémica obliga, más si cabe, a aunar esfuerzos para avanzar en la sociedad y la economía del conocimiento. El...
Premiadas dos tecnologías del Banco de Patentes, se presentarán en Bio Boston
Dos tecnologías del Banco de Patentes, Sistema de Sellado Epidural y Antisense RNA-therapeutics in Myotonic Dystrophy, entre las ganadoras del Programa de Aceleración Comercial Internacional FIPSE...
Back ENDOFIS
ENDOFIS
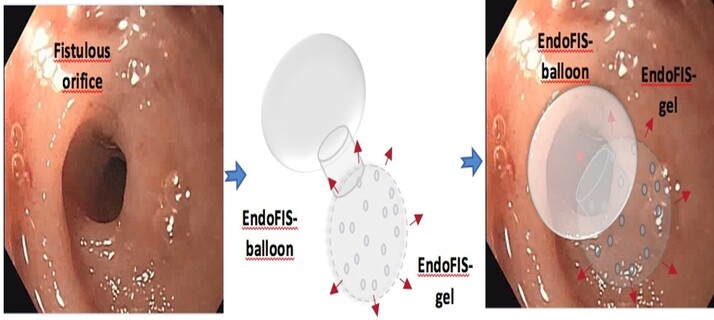
Endoscopic treatment is the first choice in the management of these post-surgical complications, as it is a less invasive and more cost-effective technique than surgical re-intervention. The endoscopic techniques available today are based on mechanical methods, such as clips, covered expandable prostheses, vacuum therapies, vacuum systems or tissue sealants, whose clinical efficacy is low, less than 50%, as technical success is not always achieved for various reasons (poor manoeuvrability, failure in anchorage or migration of the sealing fluid, etc.).
For this reason, a new medical device has been developed as we have a clear unmet need in this field, therefore there is a strong niche market for our biomedical device.
EndoFIS is a class III medical device, biocompatible and biodegradable, which allows a novel combined therapeutic approach, both mechanical and bioactive through a mechanical closure system using a double porous biodegradable balloon, made with a combination of different materials, which allows the closure of the post-surgical fistula by means of a system of approximation and fixation to the wall of the digestive tract around the fistulous orifice (EndoFIS balloon); and another bioactive by release of sealing hydrogels through the pores of the balloon, which can also function as a platform for the release of specific drugs for each patient (EndoFIS gel).
The advantages of this product are unquestionable, as we believe that it will substantially increase the clinical efficacy of the systems currently available, considerably reducing morbidity and mortality and the costs associated with this complication, the incidence of which is increasing given the large increase in surgical procedures, promoting an efficient health and healthcare model by stimulating private investment based on innovation and experimentation.
The product, co-owned by IIS La Fe and UPV-CBIT, has been registered as a European Patent with application number EP22382100, and application date February 7, 2022.
Patent title: ''Fistula sealings''.
OTRI IIS La Fe
otri@iislafe.es
+0034 618 73 00 95
Health Research Institute Hospital La Fe
Avinguda de Fernando Abril Martorell, nª 106
46026 Valencia- SPAIN